International Vaccine Institute (IVI) visits SCARLETRED Headquarters in Vienna

The International Vaccine Institute (IVI) visited the SCARLETRED Headquarters in Vienna. The joint meeting highlighted the importance of companionship and collaboration for safety, efficacy and affordability of vaccines.
Founded in 1997 on the initiative of the United Nations, the International Vaccine Institute (IVI) follows the main goal to ensure the development and provision of safe, effective and affordable vaccines worldwide.
Headquartered in Seoul, IVI has branches worldwide. The institute has recently settled in the heart of Europe, having opened an office in Vienna, Austria. The opening marks an important step towards ensuring safety, efficacy and tolerability of vaccines across continents as well as enhancing collaboration opportunities with Austrian Key Players in MedTech. Across the globe, IVI has registered members in more than 40 countries.
IVI’s current portfolio includes vaccines in multiple stages of pre-clinical and clinical development targeting infectious diseases that significantly impact low- and middle-income countries, including cholera, typhoid fever, chikungunya, shigellosis, schistosomiasis, hepatitis E, HPV- and Group A streptococcal diseases, as well as COVID-19.
IVI visits SCARLETRED HQs
As a celebration of their office opening, IVI has visited the SCARLETRED Headquarters – represented by Dr. Kerstin Westritschnig, Head of the IVI Austria Country Office, alongside Colin McCann, External Affairs Manager at the IVI Europe Regional Office. They were welcomed by Dr. Harald Schnidar, CEO, Founder & President of SCARLETRED, as well as Kathrin Schnidar, CFO of SCARLETRED.
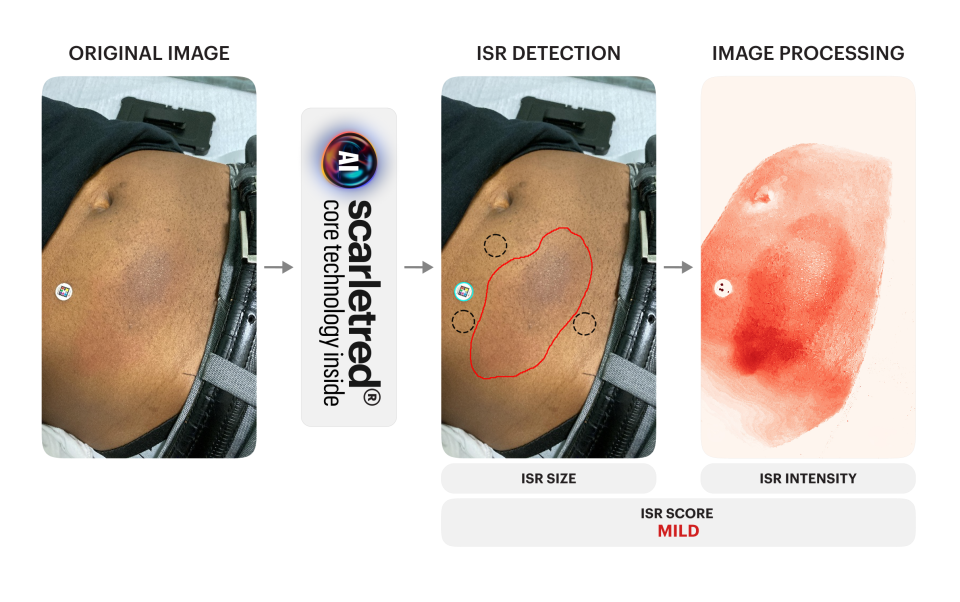
Why ISR monitoring faces global demand
The meeting represents an important steppingstone toward the joint effort to enhance safety of vaccines worldwide. The pioneering work of IVI is at the crossroads of SCARLETRED’s leading position in standardized mobile skin imaging and AI supported analysis of Injection Site Reactions (ISRs).
The U.S. Food and Drug Administration (FDA) as well as the European Medicines Agency (EMA) focus on assessing, reporting, and mitigating injection site reactions (ISRs), particularly for biopharmaceuticals, which often cause injection site pain (ISP), erythema, and swelling.
In the meeting, both organizations discussed how SCARLETRED’s capabilities could be integrated into next-generation vaccine trials to further de-risk the evaluation and development of novel vaccination therapies in line with the latest FDA and EMA guidance.
State of the art research on ISR monitoring and AI-powered detection
ISRs have been an intensely discussed topic, backed up by scientific publications. As a pioneer in clinically validated skin imaging and monitoring, SCARLETRED has published longitudinal research on injection site reaction monitoring, AI-powered detection and inclusive quantification.
Download the full paper here.
AI-powered ISR monitoring with Scarletred® Vision
Injection Site Reactions (ISRs) are common adverse effects of injectable treatments, negatively impacting patient comfort, and overall treatment outcomes. SCARLETRED shows pioneering work in ISR monitoring:
The AI-enhanced, mobile imaging solution Scarletred® Vision enables remote, patient-driven documentation, proactive monitoring, and timely clinical intervention, supporting proactive care and reducing the need for clinic visits.
Scarletred® Vision is accessible worldwide on any smartphone and across multiple trial sites, regardless of setting or continent. For clinicians, automated quantification provides consistent, objective data that can be seamlessly integrated into existing workflows, improving comparability across sites, and over time.
Aligned vision for accessible healthcare worldwide
SCARLETRED was honored to host IVI at its headquarters and sees strong alignment with IVI’s mission to improve safety, accessibility and global reach of healthcare solutions. The parties look forward to potential collaboration aimed at advancing the safe, efficient, and globally accessible development of vaccines through the medical device-certified Scarletred®Vision system for digital skin assessment and monitoring.


